It follows the clearance for connecting Hailie with AstraZeneca’s Symbicort inhaler.
Adherium gets US FDA 510(k) to connect remote monitoring sensor for Ellipta inhaler users
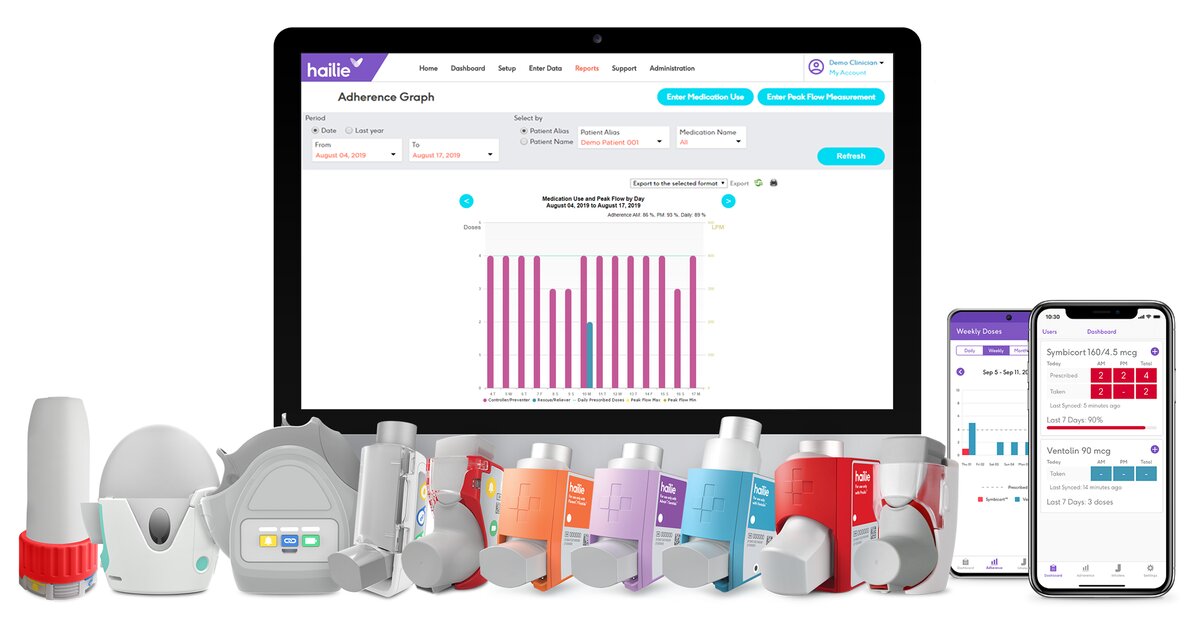

It follows the clearance for connecting Hailie with AstraZeneca’s Symbicort inhaler.